28 reports that in 21 out of 31 PNH patients on eculizumab the direct antiglobulin test (DAT) was positive for C3 on red cells (one was also weakly positive for IgG), whereas only one had been DAT-positive before treatment in contrast, out of 39 PNH patients not on eculizumab only 2 were DAT-positive for C3. 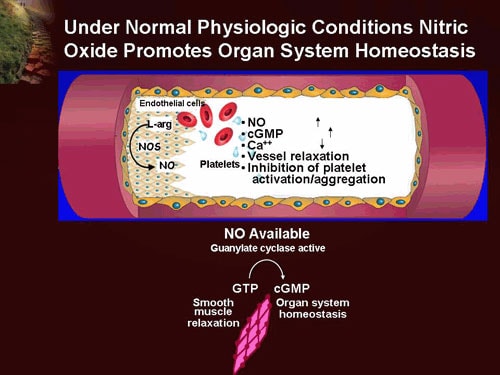
However, the data provide evidence that in patients on eculizumab there are changes in objective parameters that may correlate with the risk of thrombosis, and this may also help to unravel the puzzle of thrombophilia in PNH. These findings do not resolve the issue as to whether eculizumab prevents thrombosis indirectly by curbing intravascular hemolysis (which perhaps triggers thrombosis through the release of thromboplastin-like substances), or whether it acts directly by blocking C5 (which in turn might activate platelets and/or the coagulation pathway) 30. have systematically investigated patients on eculizumab and they have found that, compared to pre-treatment values, they tend to have lower levels of coagulation factors such as von Willebrandt factor, of by-products of coagulation such as D-dimers, and also of endothelial cell molecules such as VCAM. In this issue of Haematologica, a paper by Gerard Socié’s group 27 deals with how eculizumab may influence venous thrombosis in PNH, and Peter Hillmen’s group provide data 28 on the recently developed notion 29 that PNH patients on eculizumab show evidence of extravascular hemolysis, which may be an important determinant of the clinical response to this new therapy. 25 In view of this, it has come as an added bonus that patients on eculizumab therapy have a substantially lower risk of thrombosis. 5, 22– 24 On the other hand, if all patients with PNH are given anticoagulant prophylaxis the added burden is significant: in addition, it is unnecessary for one-half of them, it does not completely prevent thrombosis, and it entails the risk of serious hemorrhage. If a patient with PNH is not given anti-coagulant prophylaxis, there is a significant risk that sooner or later that patient may develop a serious thrombotic complication. Until we know the answers to these questions, they pose a difficult management problem. 21 There are two main questions: (a) what triggers a major thrombotic event in an individual patient, sometimes years into the course of the disease (b) why are about one-half of the patients spared this complication. PNH is associated with the most vicious acquired thrombophilic state known to medicine, with nearly one-half of untreated patients sooner or later affected: the consequences are potentially devastating, 5, 18– 20 and it is humbling to admit that we still don’t know the mechanism. But the idea has become a reality through the advances of biotechnology, when Alexion developed a humanized anti-C5 monoclonal antibody, eculizumab, 11 which has proved highly effective in the control of intravascular hemolysis in patients with PNH: 12– 14 a huge step forward in the treatment of disorder that until now could be cured only by hematopoietic stem cell transplantation. 
9, 10 Therefore stopping hemolysis by blocking complement was a logical idea: it did not seem easy to achieve this safely in vivo. 
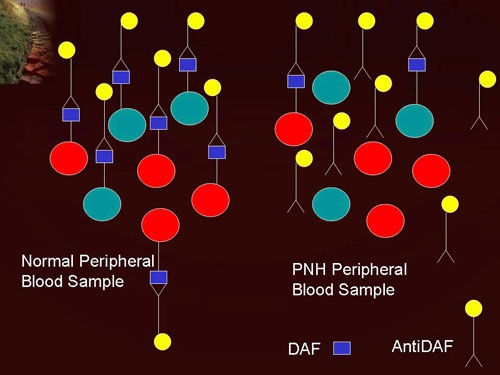
That in PNH hemolysis was complement-dependent has also been known for a long time ( Figure 1): indeed, the acidified serum (or Ham-Dacie) test, based on complement-mediated lysis of PNH cells in vitro, has for half a century been the gold standard for the diagnosis of PNH, 7, 8 until flow cytometry came into its own. 4 The last qualification is not trivial, because even today the accesses of hemoglobinuria, most distressing for the patient, may tend to obscure the fact that in PNH hemolysis goes on all the time: macroscopic hemoglobinuria merely reflects its exacerbations. 1– 3 But in this journal, founded by Adolfo Ferrata in the twenties, it seems appropriate to remember that, in the Italian literature, the full name coined for PNH in 1928 by Ettore Marchiafava was even more articulate: paroxysmal nocturnal haemoglobinuria with perpetual haemosiderinuria. Paroxysmal nocturnal hemoglobinuria (PNH) is enough of a tongue-twister for patients and doctors alike and we all teach the students that PNH is a disorder characterized by the triad of intravascular hemolysis, venous thrombosis and cytopenias. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
